By Lexico Health – Mogadishu, Somalia
MOGADISHU, Somalia – In a year dominated by humanitarian pressures, rising health needs and fragile infrastructure, one Somali-born pharmaceutical manufacturer continued to quietly shift the country’s medical landscape.

Yenisom Healthcare – recognized as the first private pharmaceutical manufacturing factory in the history of the Somali Republic – used 2025 to consolidate its position as a cornerstone of Somalia’s emerging health economy, expanding its product range, deepening institutional ties and sharpening its role in national health resilience.
From its production site along KM4, Tarbuunka Road in Mogadishu’s Hodan District, the company now manufactures a growing list of essential medicines, including tablets, syrups and injectables, aimed at both domestic and regional markets.
Building on a Vision Born in Crisis

Yenisom Healthcare – recognized as the first private pharmaceutical manufacturing factory in the history of the Somali Republic
Founded on 28 February 2022, Yenisom was conceived as a response to a long-standing gap: Somalia’s heavy dependence on imported medicines and the absence of a modern, locally regulated production base.
In the words of its CEO, Professor Cabdi Aakhiro, Yenisom was designed as “a catalyst for change,” blending Somali and Turkish pharmaceutical expertise to advance Africa’s pharmaceutical industry starting from Somalia.
That vision remained the guiding theme of 2025. While global and national attention often focused on emergency needs – drought, displacement, disease outbreaks and a health system under strain – Yenisom spent the year reinforcing a quieter but foundational pillar: sustainable, regulated, locally produced medicine.
Expanding the Product Portfolio
One of the key storylines of 2025 for Yenisom was the ongoing expansion and visibility of its product portfolio across the Health & Medicine category. The company’s branded generics now include formulations such as:
- Yeniprofen (Ibuprofen 400 mg) – for pain and inflammation
- Yenixiume 40 mg (Esomeprazole tablet) – for gastric acid-related conditions
- Yenimol 500 mg – a paracetamol-based analgesic
- Yenisilin 500 mg and 1000 mg – antibiotic preparations
- Yenigyl, Yenithyromycine 250 mg, and Cintrrom multivitamin – addressing infections and nutritional gaps.
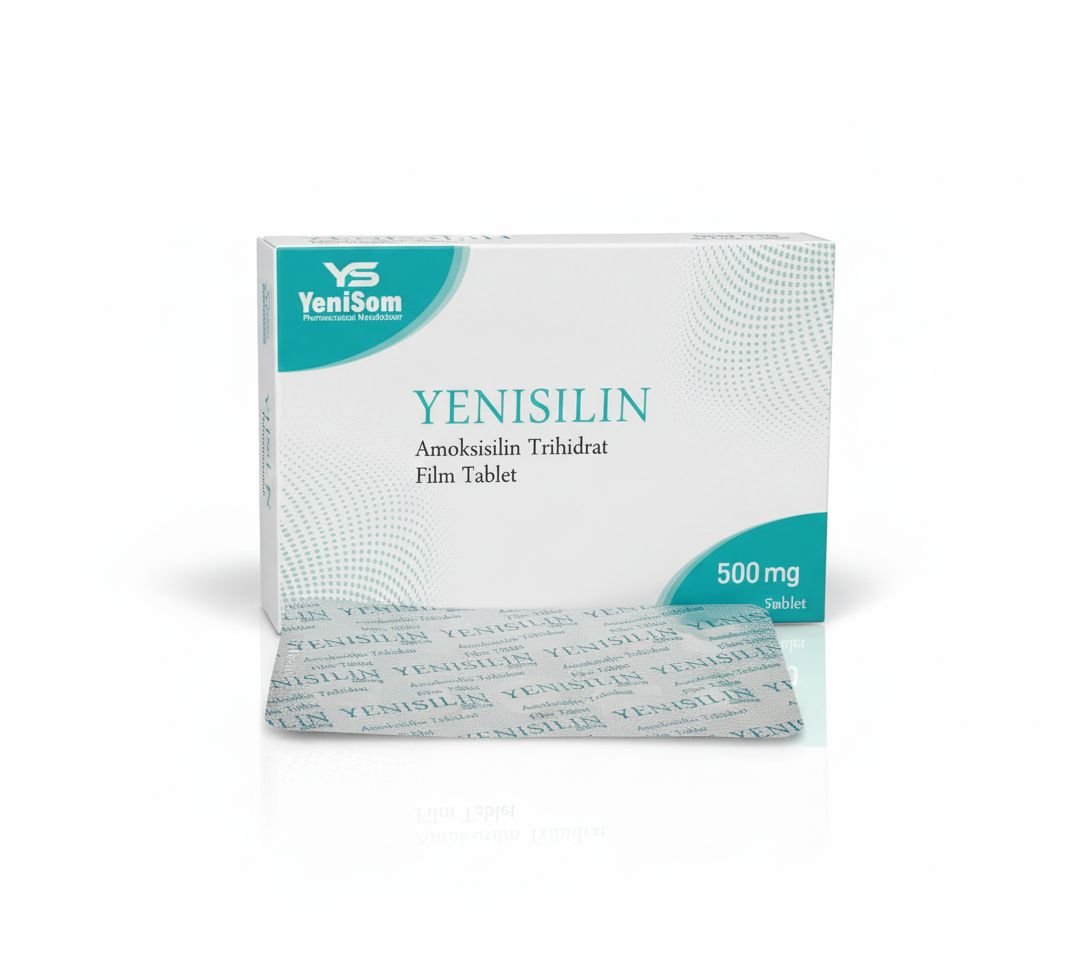


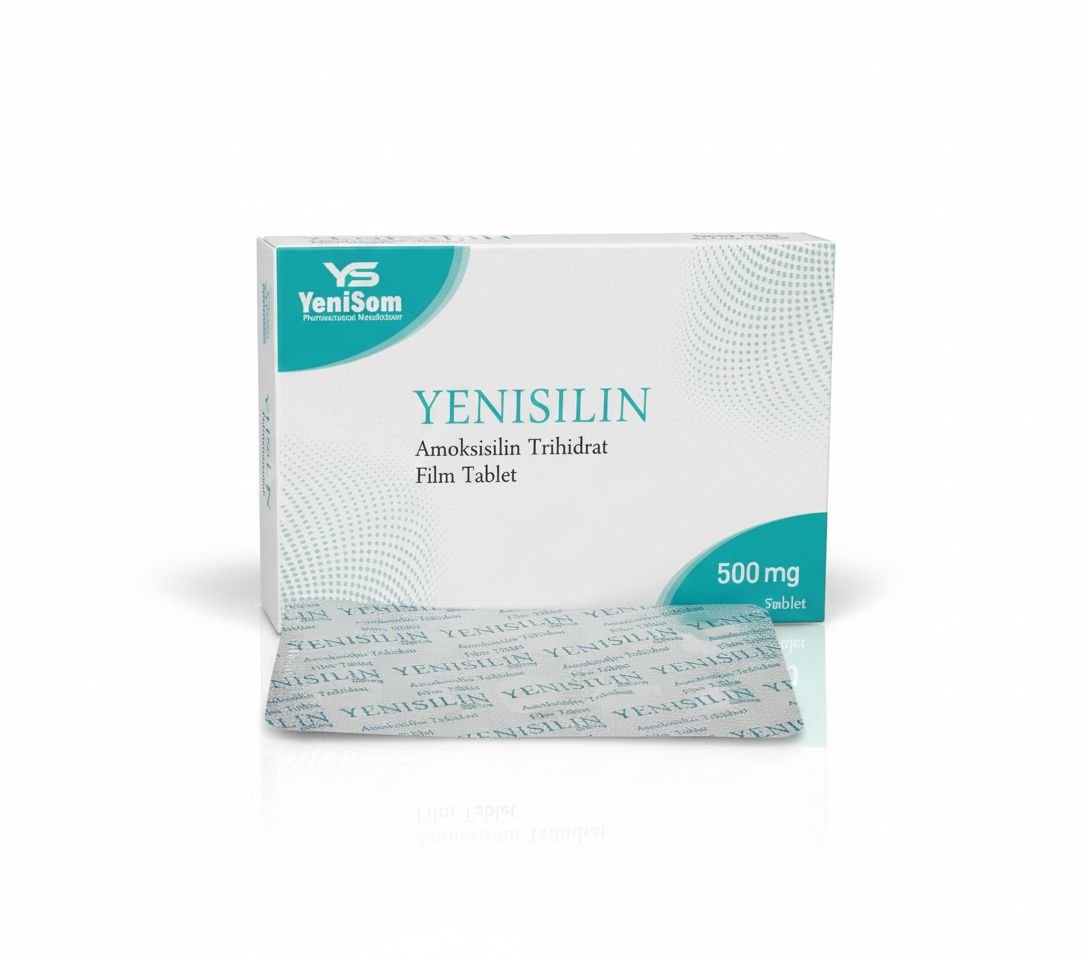

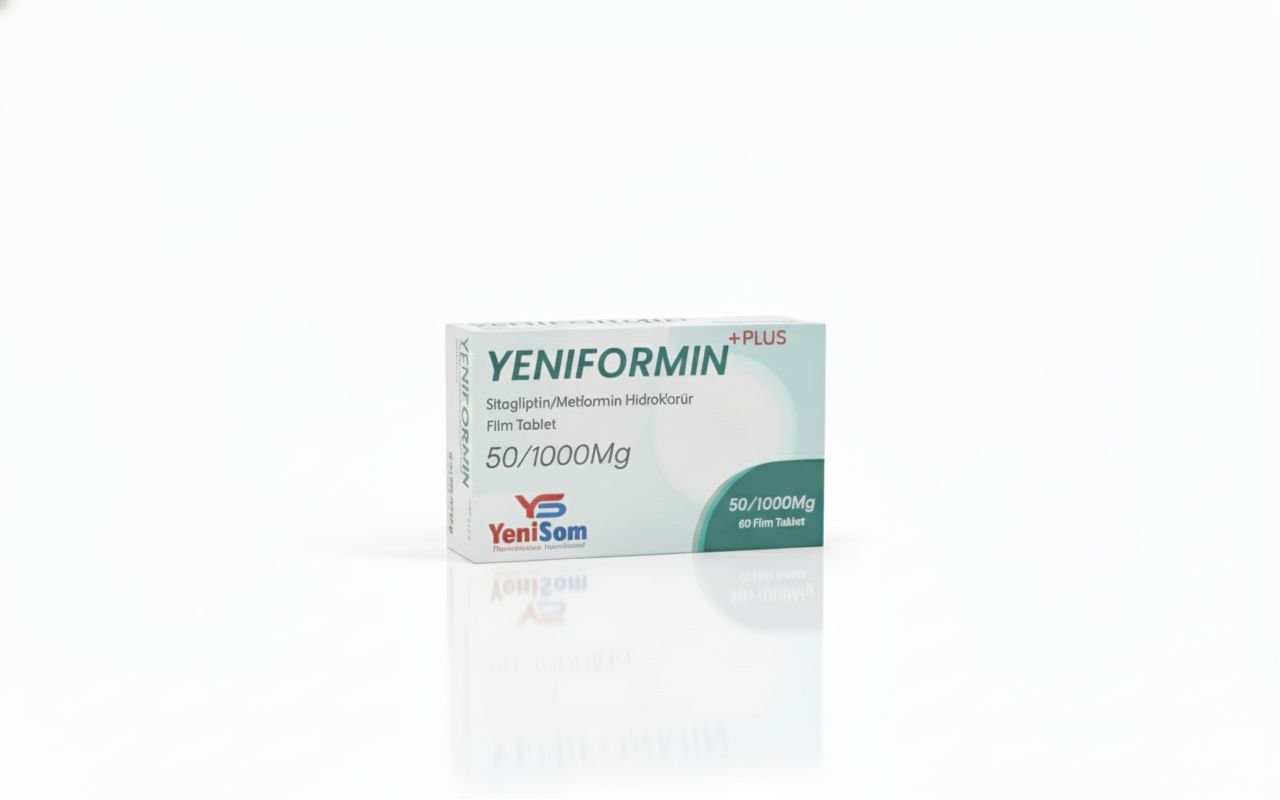

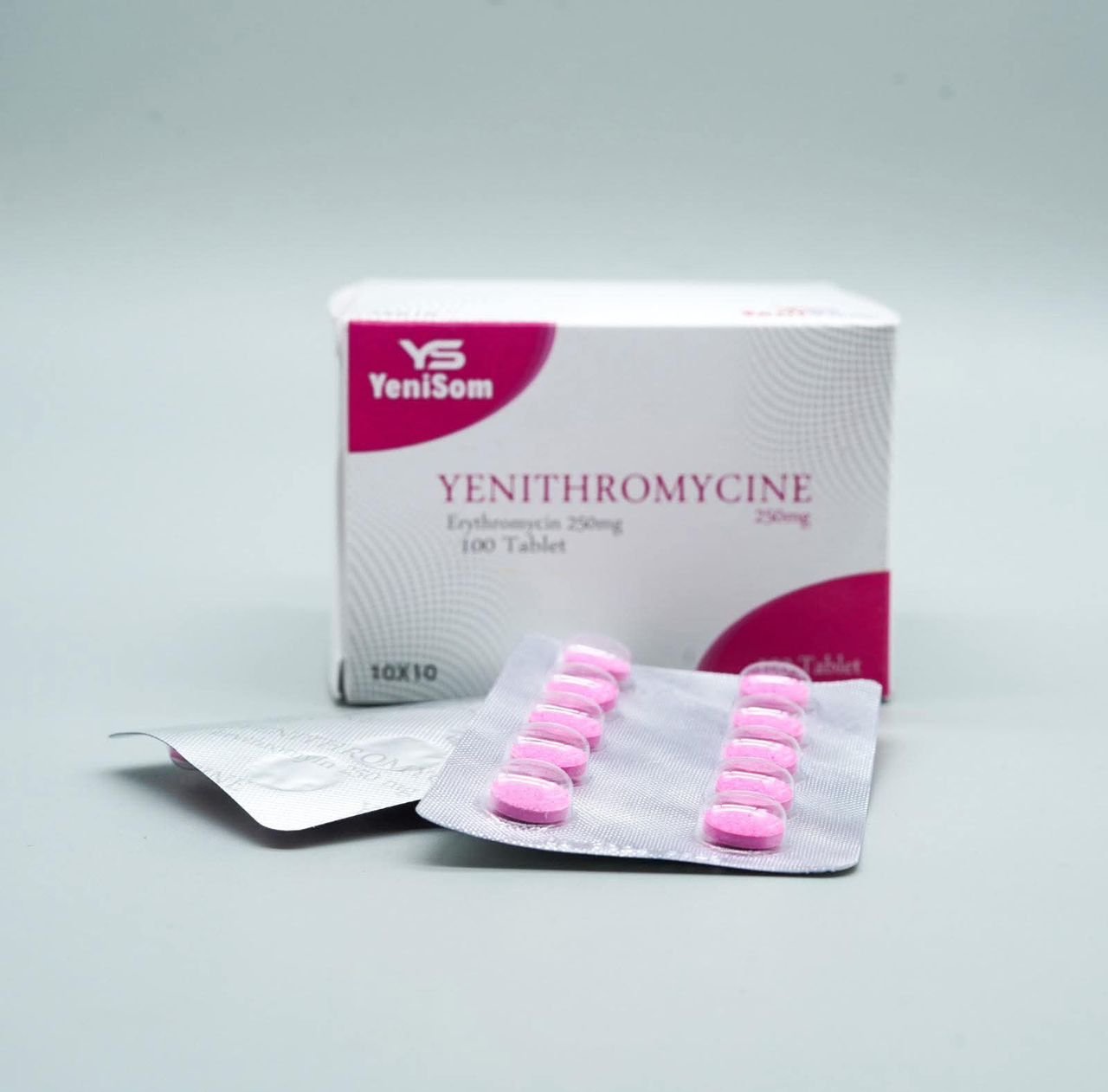

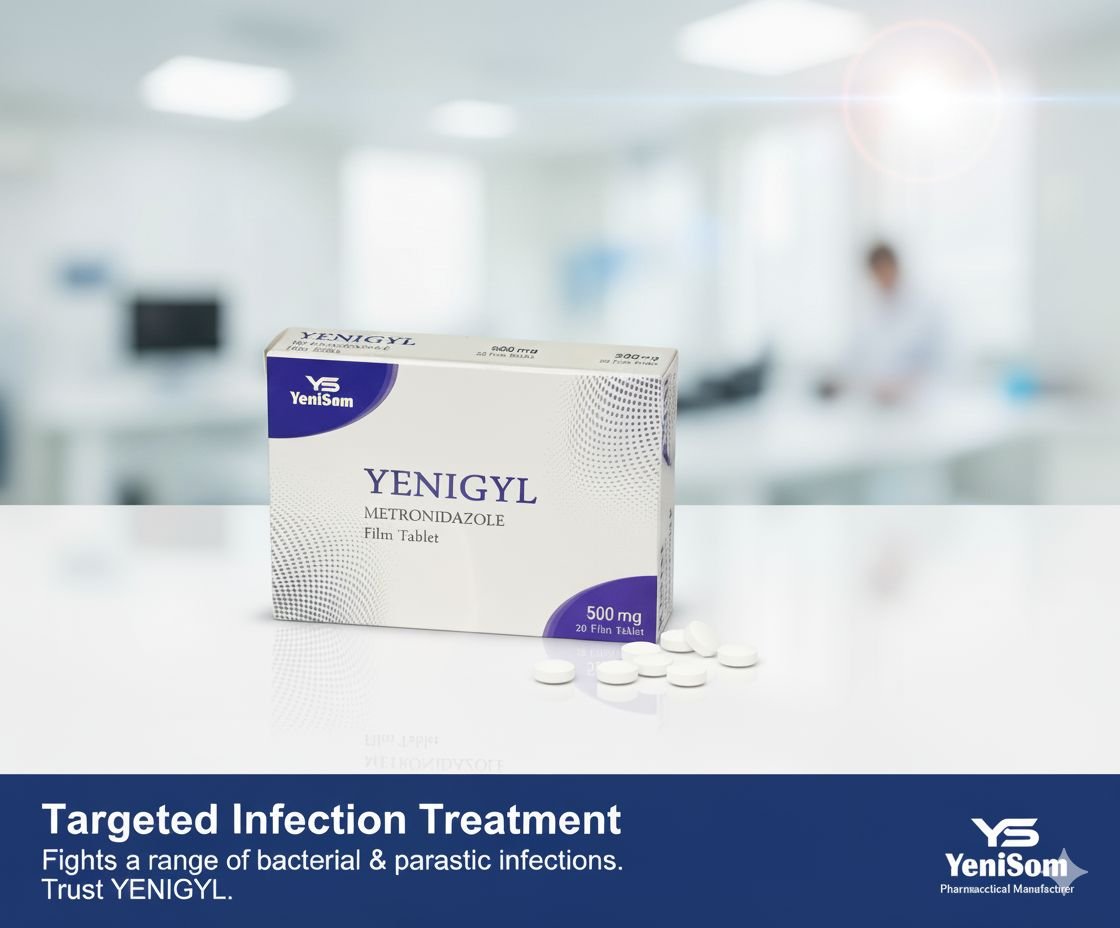
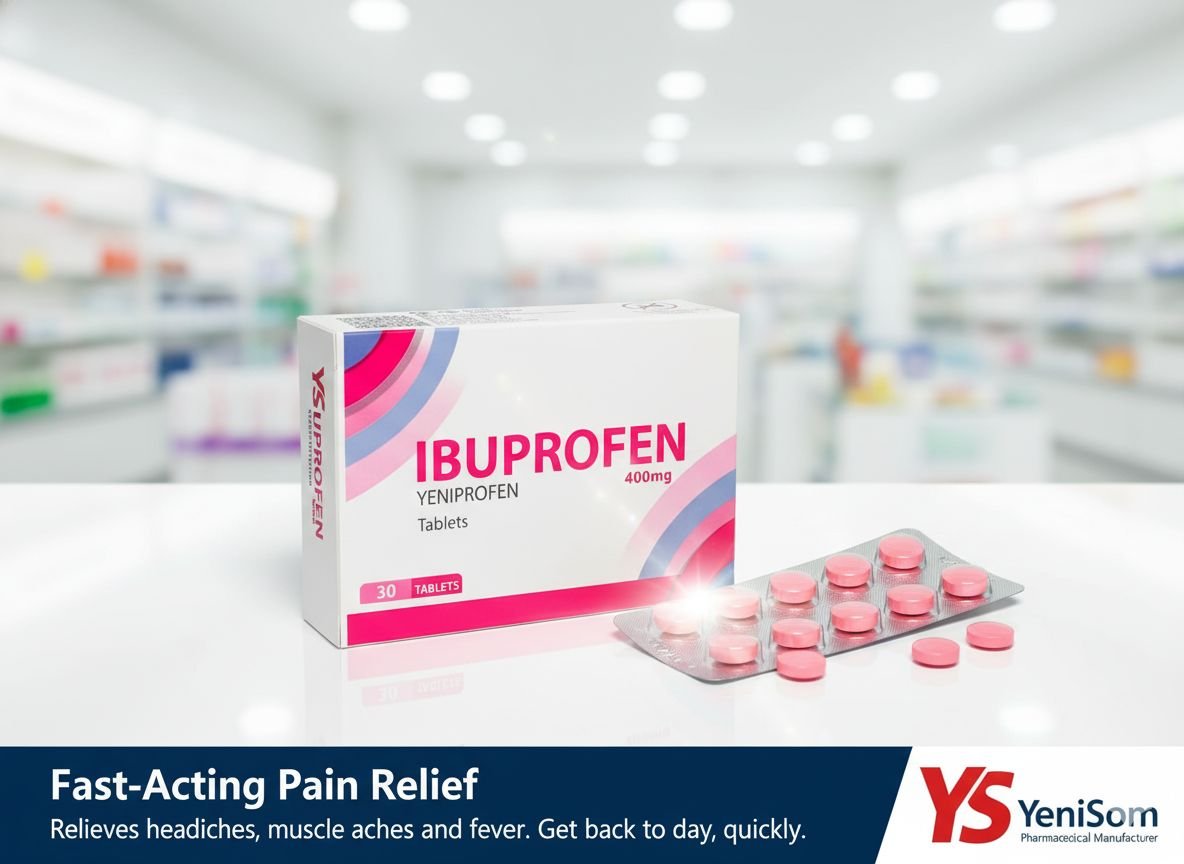




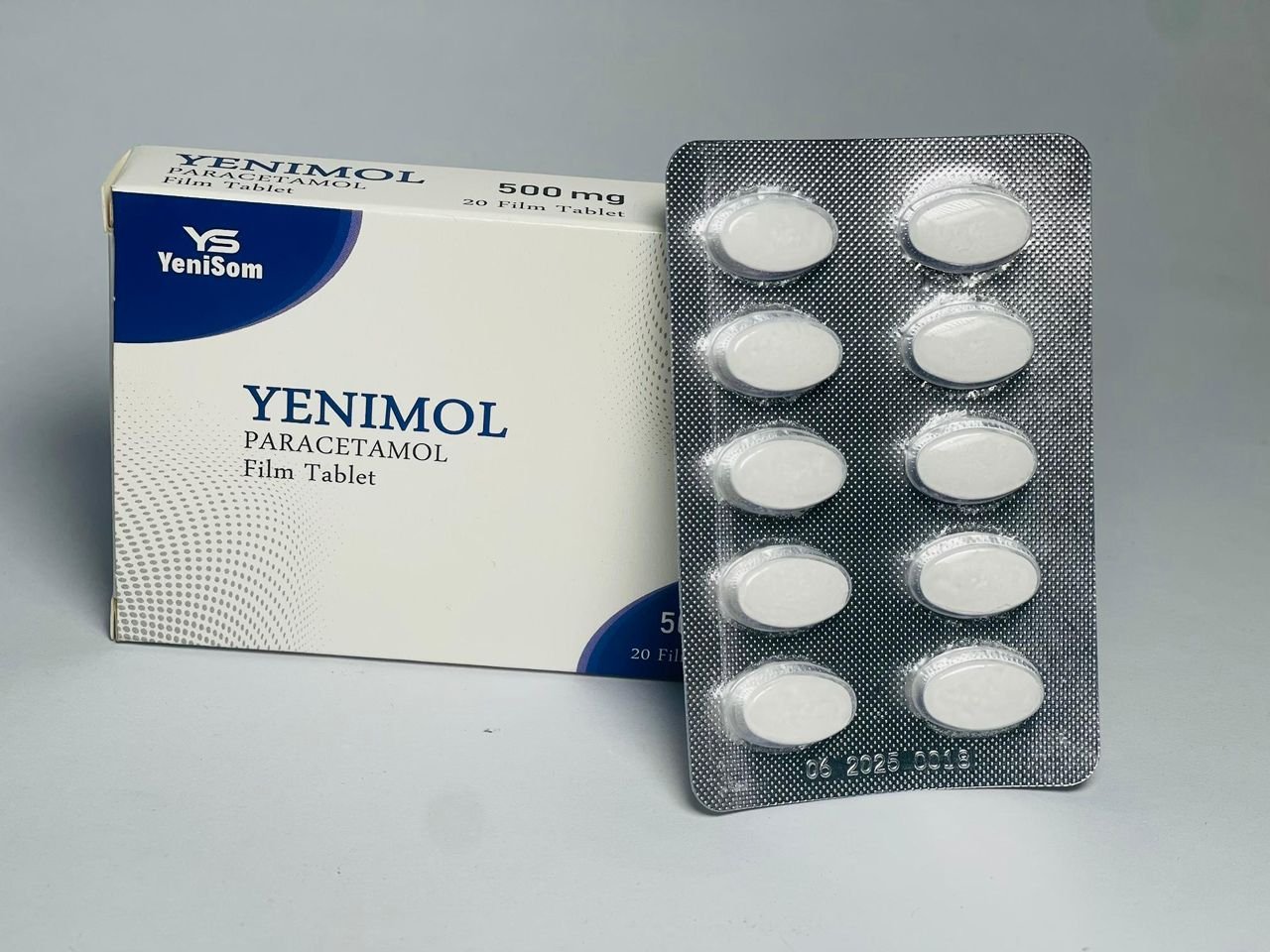
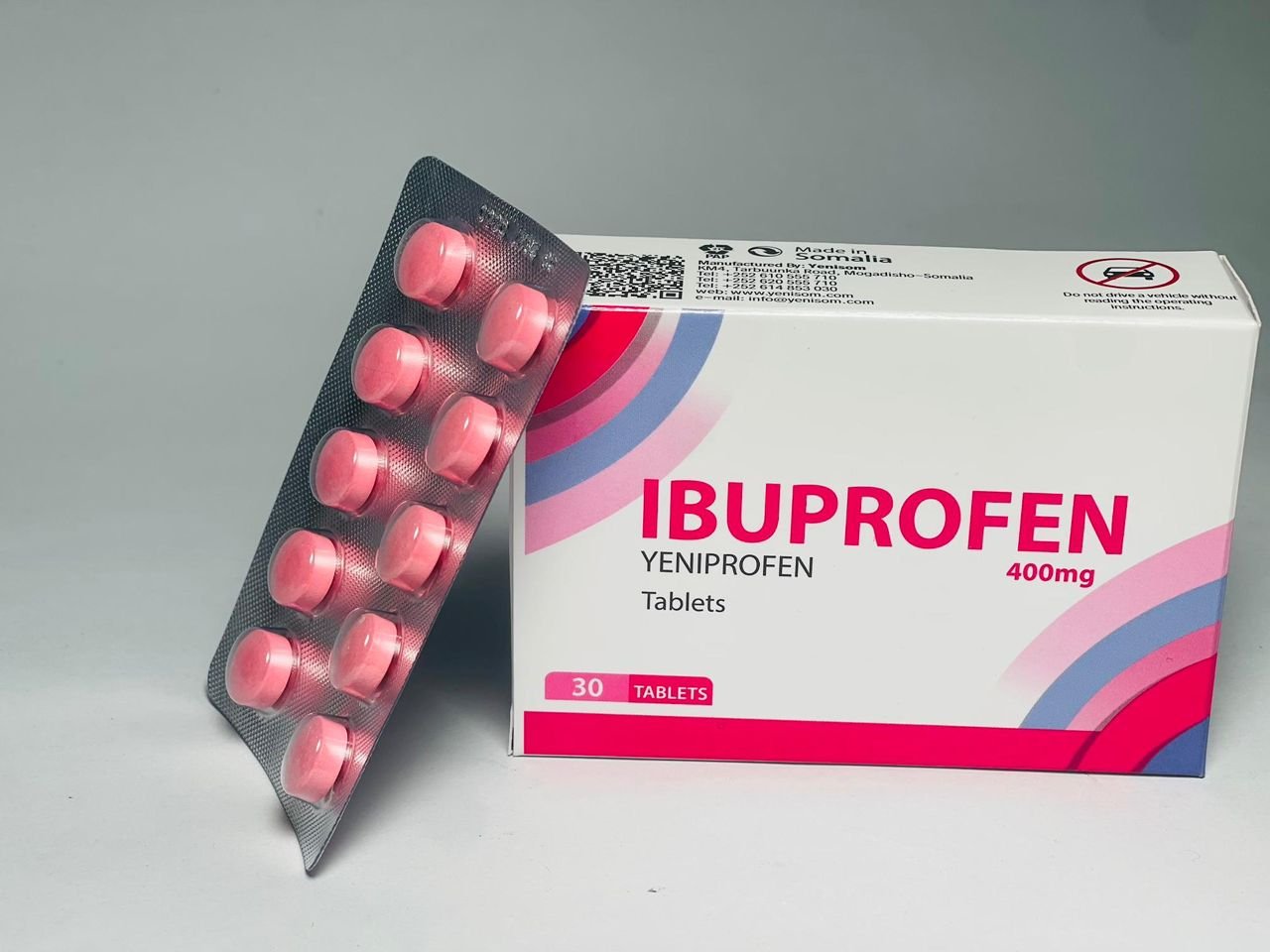
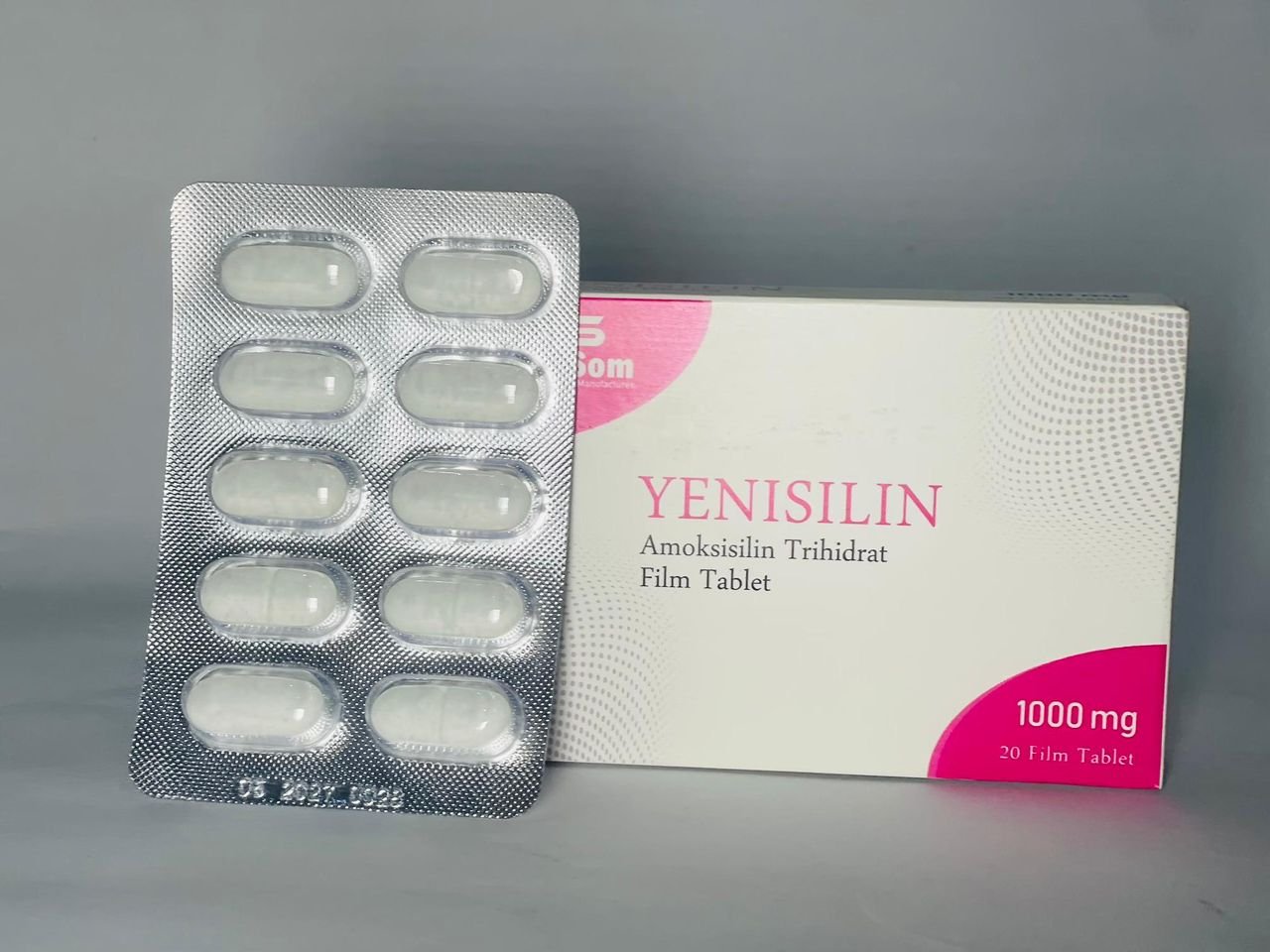
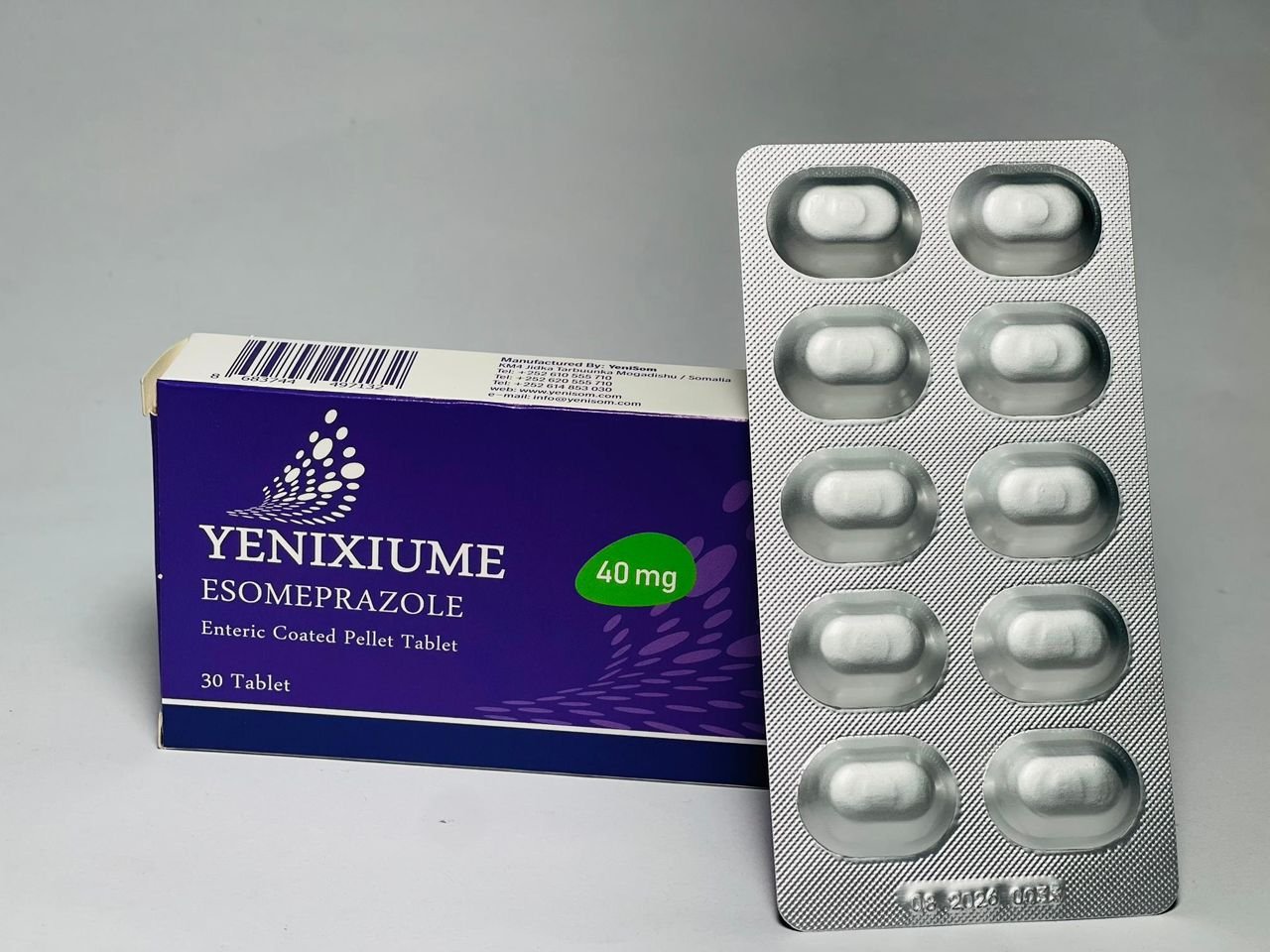
These products, manufactured in Mogadishu, are marketed as quality-assured and affordable options for pharmacies, clinics and hospitals, offering a critical alternative to often expensive or inconsistently supplied imports.
The expansion of the product line in 2025 aligned with the company’s stated mission: to improve access to high-quality generic medicines for communities across East Africa, while anchoring production capacity inside Somalia itself.
UNICEF Visit: Local Capacity Meets Global Mandate

Although the visit itself took place in October 2024, its implications reverberated well into 2025 and formed a central pillar of Yenisom’s public narrative this year.
On 21 October 2024, Mr. Tom Ziraguma, Chief of Supply and Logistics for UNICEF Somalia, visited the Yenisom Pharmaceutical Site in Mogadishu. The visit – later profiled on Yenisom’s news page in February 2025 – focused on assessing the facility’s capacity to support ongoing health and humanitarian needs nationwide.
During the tour, UNICEF representatives examined production lines, storage, quality control procedures and overall readiness to supply medicines at scale. The interest of a major global agency in a Somali private manufacturer marked an important recognition: that domestic production has a real role to play in a country where international organizations traditionally source supplies from abroad.
Against the backdrop of UNICEF’s 2025 humanitarian appeal – which seeks US$171 million to support 1.8 million people in Somalia with integrated health, nutrition and WASH services – the availability of a local pharmaceutical partner strengthens prospects for faster, more cost-effective supply chains inside the country.

While no formal long-term supply agreement has been publicly announced, the visit alone signaled that Yenisom is increasingly viewed as part of the broader health-response ecosystem rather than an isolated private venture.
Mr. Tom Ziraguma, Chief of Supply and Logistics for UNICEF Somalia, visited the Yenisom Pharmaceutical Site in Mogadishu.
Yenisom’s 2025 story unfolds against a wider national context that remains deeply challenging.
Somalia faces overlapping crises: climate shocks, displacement, conflict-related insecurity and persistent disease outbreaks. UNICEF estimates that in 2025, 6.9 million people in Somalia require humanitarian assistance, including 4.3 million children, with hundreds of thousands of young children at risk of acute malnutrition.
Health-sector needs remain acute: millions require health support and access to essential medicines, while outbreaks of cholera and vaccine-preventable diseases continue to threaten already stretched communities.
In this environment, Yenisom’s value proposition is straightforward but significant:
- Shorter supply lines – Manufacturing inside Mogadishu reduces dependency on complex import logistics.
- Better alignment with local needs – A Somali-based producer can respond more quickly to shifts in disease patterns and demand.
- Economic co-benefits – Jobs in production, quality control, distribution and administration contribute to livelihoods in a country where employment is scarce. This combination of health and economic impact is why analysts increasingly view domestic pharmaceutical manufacturing as a strategic sector for Somalia’s long-term resilience.
Operating in a High-Need, High-Risk Environment
Quality, Regulation and Trust


A key thread running through Yenisom’s communications in 2025 has been the emphasis on quality assurance and regulatory compliance.
The company is authorized and regulated by the Ministry of Commerce and Industry and operates in alignment with national health regulations, with standards certified by the Somali Bureau of Standards (SBS).
Its facilities encompass manufacturing lines, research and development, quality-control laboratories, quality-assurance units and storage environments designed to protect medicine integrity.





In a market where public confidence in medicine quality can be undermined by counterfeit or substandard imports, Yenisom is positioning itself as a trustworthy local alternative – blending international quality benchmarks with local accountability.
Yenisom’s trajectory during 2025 also carries symbolic weight.
For decades, Somalia’s health sector has relied heavily on external suppliers and international donors. A domestic company that is:
- founded and led by Somali professionals,
- partnering with Turkish pharmaceutical expertise,
- employing local pharmacists, chemists and technicians, and
- building Somali medicine brands represents a tangible form of local ownership over a critical part of the health system.
For younger Somali health professionals and entrepreneurs, Yenisom functions as both employer and example: proof that high-value industrial and scientific work can be carried out inside Somalia, not only abroad.
Challenges on the Road to Scale
Yet the 2025 year recap is not without its caveats. Scaling pharmaceutical manufacturing in Somalia means navigating:
- Security risks that can disrupt operations and logistics
- Infrastructure gaps such as power reliability and transport networks
- Regulatory capacity constraints, as national institutions still rebuild after years of conflict
- Market volatility, with fluctuating demand and purchasing power across regions
These structural obstacles, widely documented in humanitarian and development reports on Somalia, form the backdrop to Yenisom’s
operations and limit how quickly domestic production can meet the full breadth
of national needs.
Company leadership has consistently framed these hurdles not as deterrents but as the very reason Yenisom exists – to demonstrate that sophisticated, standards-compliant manufacturing is possible even in a fragile state.
As 2025 draws to a close, Yenisom’s outlook is increasingly regional.
The company’s founding mandate was not only to meet Somalia’s needs but to contribute to pharmaceutical supply across East Africa. With product lines already organized under health and medicine categories and a growing list of branded generics, Yenisom is positioning itself for broader export opportunities where regulatory frameworks and logistics permit.


At the same time, partnerships with multilateral actors, humanitarian agencies and national health authorities remain a central strategic priority – not only as potential clients, but as collaborators in improved medicine access, rational drug use and health-system strengthening.
A Year That Consolidated a Quiet Revolution
In purely commercial terms, 2025 may be remembered as a year of consolidation rather than dramatic expansion for Yenisom Healthcare: refining its product range, strengthening its brand, deepening regulatory relationships and demonstrating its capacity to stakeholders like UNICEF.
In broader national terms, however, the company’s work forms part of a quiet revolution: the slow but crucial shift from a fully import-dependent medicine market to one where Somalia can produce, regulate and trust its own pharmaceuticals.






























Leave a comment